 :max_bytes(150000):strip_icc()/PeriodicTable-56a12c983df78cf772682271.png)
Thus, the formula mass of calcium hydrogen carbonate is 117.10 amu and the molar mass of calcium hydrogen carbonate is 117.10 grams per mole (g/mol). However when talking about a mole of an ionic compound we will still use the term molar mass. This is because there are no individual molecules in ionic compounds. For compounds that are not molecular (ionic compounds), it is improper to use the term “molecular mass” and “ formula mass” is generally substituted. The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) = 28.02 amu. Determining the Molar Mass of a Molecule(opens in new window) youtu.be The molar mass of naturally-occurring carbon is different from that of carbon-12, and is not an integer because carbon occurs as a mixture of carbon-12, carbon-13, and carbon-14. For a molecule (for example, nitrogen, N 2) the mass of molecule is the sum of the atomic masses of the two nitrogen atoms. The periodic table lists the atomic mass of carbon as 12.011 amu. The concept of molar mass can also be applied to compounds. Further, if you have 16.00 grams of oxygen atoms, you know from the definition of a mole that your sample contains 6.022 x 10 23 oxygen atoms. For example, the atomic mass of an oxygen atom is 16.00 amu that means the molar mass of an oxygen atom is 16.00 g/mol. It can supply you with an exact representation of a substance’ssize and mass, and valence electron shell. Generalizing this definition, the molar mass of any substance in grams per mole is numerically equal to the mass of that substance expressed in atomic mass units. Periodic Table With Atomic And Molar Mass The Periodic Dinner table is an essential part of the research into scientific research, also it can be helpful in figuring out a substance’s attributes. 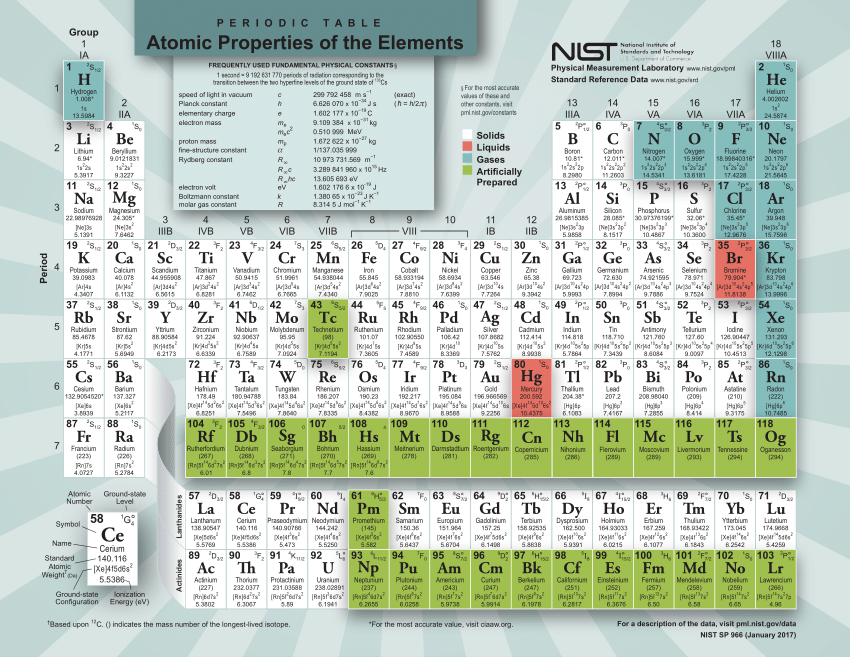
By definition, the molar mass of carbon-12 is numerically the same, and is therefore exactly 12 grams. We have defined a mole based on the isotopic atomic mass of carbon-12. The atomic mass of an element is the relative average of all of the naturally occurring isotopes of that element and atomic mass is the number that appears in the periodic table. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.) a carbon atom with six proton and six neutrons in its’ nucleus, surrounded by six electrons. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). 
For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. This list contains the 118 elements of chemistry. Design Copyright © 2017 Michael Dayah (). Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects. 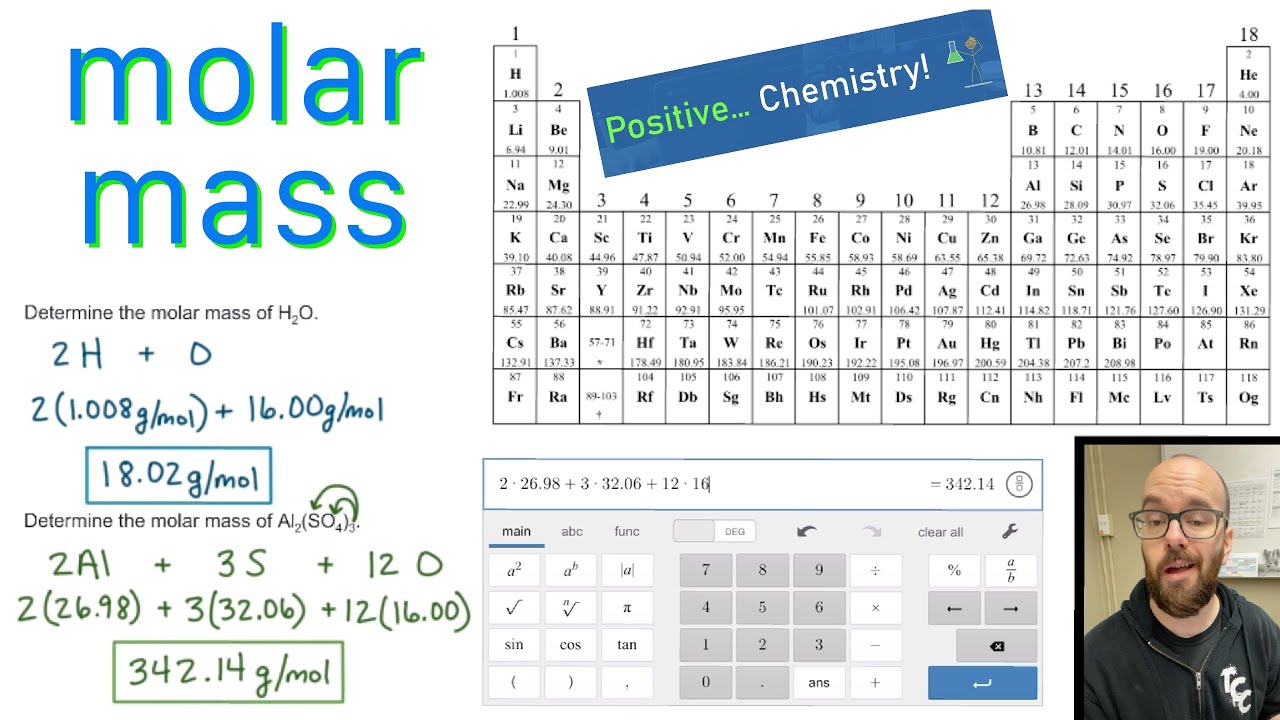
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
